
Figure 3 from Formation of hydroxylamine (NH2OH) in electron-irradiated ammonia-water ices. | Semantic Scholar

Is there a pathway for N2O production from hydroxylamine oxidoreductase in ammonia-oxidizing bacteria? | PNAS

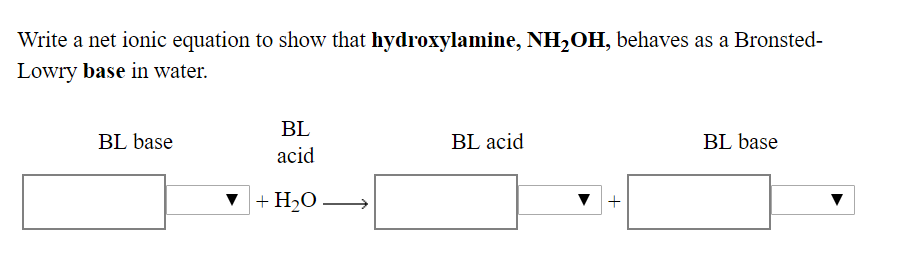
SOLVED: Write the net ionic equation to show that hydroxylamine (NH2OH) behaves as a Bronsted-Lowry base in water.

Calculate the pH of a 0.050 M solution of hydroxylamine, NH2OH. (Kb = 6.6 x 10-9) | Homework.Study.com
![PDF] Thermal decomposition of NH2OH and subsequent reactions: ab initio transition state theory and reflected shock tube experiments. | Semantic Scholar PDF] Thermal decomposition of NH2OH and subsequent reactions: ab initio transition state theory and reflected shock tube experiments. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b36782da9edaac7d0b91c7a6a1f8caff67f9661e/16-Figure22-1.png)
PDF] Thermal decomposition of NH2OH and subsequent reactions: ab initio transition state theory and reflected shock tube experiments. | Semantic Scholar

Combination of NH2OH·HCl and NaIO4: a new and mild oxidizing agent for selective oxidation of alcohols to carbonyl compounds - ScienceDirect

Scheme 2. Reagents and conditions: (i) NH2OH.HCl, Pyridine, H2O, 24 h,... | Download Scientific Diagram
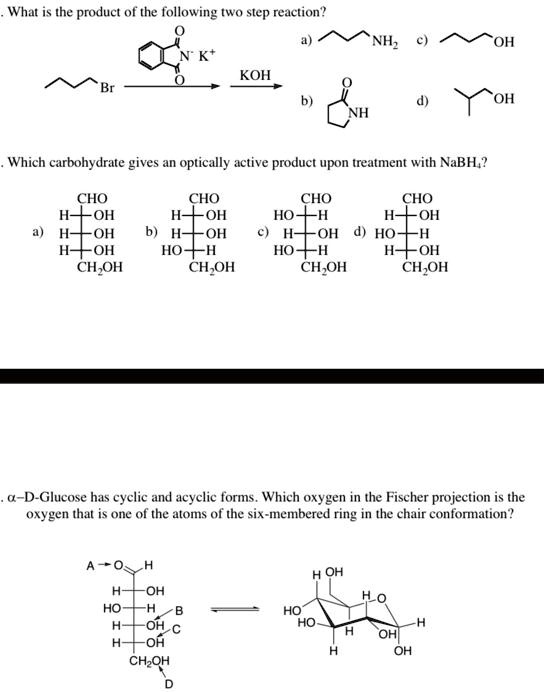
SOLVED: What is the product of the following two-step reaction? NH3 + KOH â†' NH2OH + H2O Which carbohydrate gives an optically active product upon treatment with NaBH4? CHO H-OH H-OH H-OH

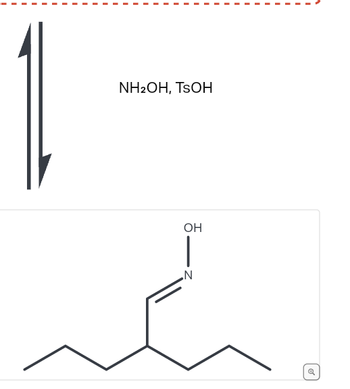





![Malayalam] In the reaction 'A+2B+H2O gives C+2D. draw the structur Malayalam] In the reaction 'A+2B+H2O gives C+2D. draw the structur](https://static.doubtnut.com/ss/web-overlay-thumb/8095007.webp)