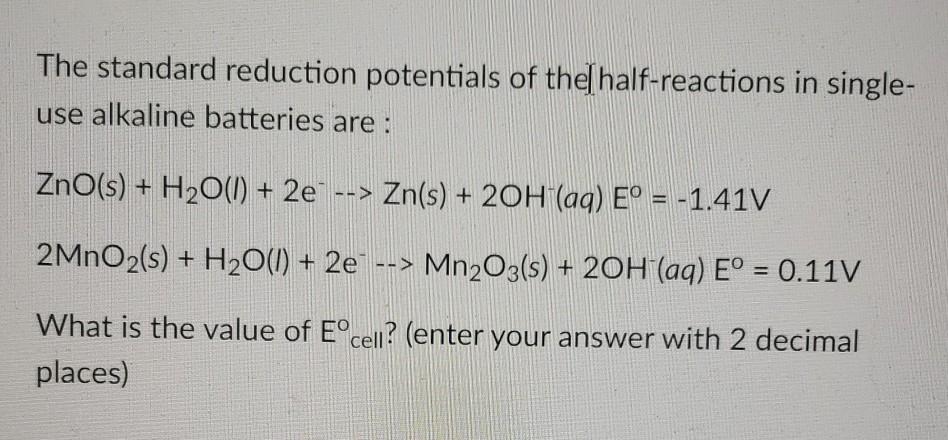Coque Liseuse Numérique Kobo Aura H2O 2e Edition Etui Ebook avec Rabat Magnétique pour Kobo - Rose foncé - Cdiscount Informatique

Ni Os(s) + H2O(0) + 2e →→ 2NO(s) + 20H"(aq) E° = 0.4V FeO(s) + H2O(1) + 2e - Fe(s) + 2OH(aq) E =-0.87 The overall reaction the cell has a value
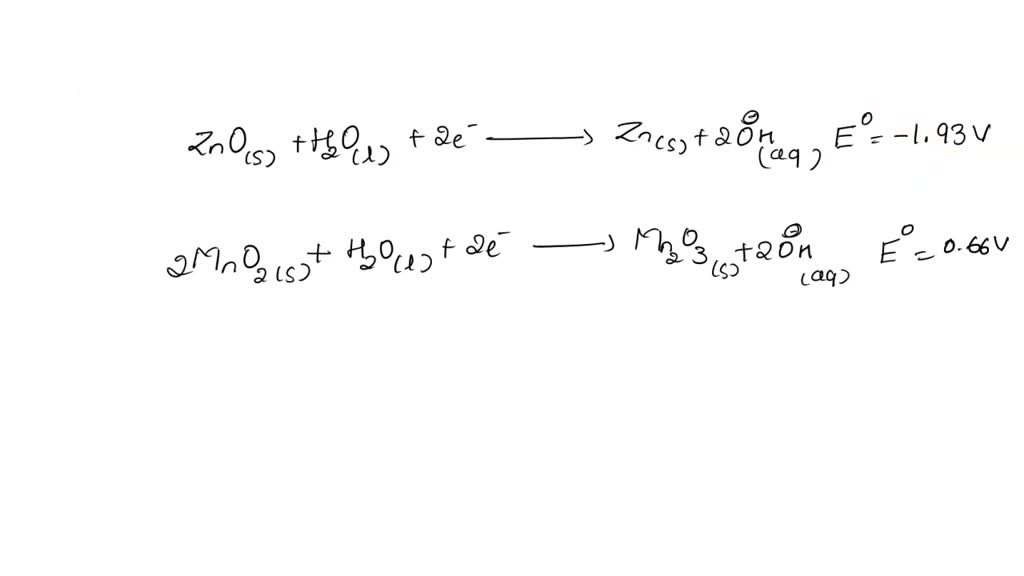
SOLVED: The standard reduction potentials of the half-reactions in single-use alkaline batteries are: ZnO(s) + H2O(l) + 2e- –> Zn(s) + 2OH-(aq) Eo = -1.93V 2MnO2(s) + H2O(l) + 2e- –> Mn2O3(s) +

E liquide Speculoos Orange Jus de Gabriel Laboratoire H2O, eliquide 50ml Orange Jus de Gabriel - Taklope
57. In acidulated water H2so4— H+ +so4 2 At cathode the reaction will be H2o +2e——h2 + oh But why donot H+ produced initially from acid compete with water to react at cathode.

Étui souple à fermeture éclair pour kobo clara/Nia/ clara 2e/libra H2O/libra 2/aura HD/aura H2O ereader - AliExpress

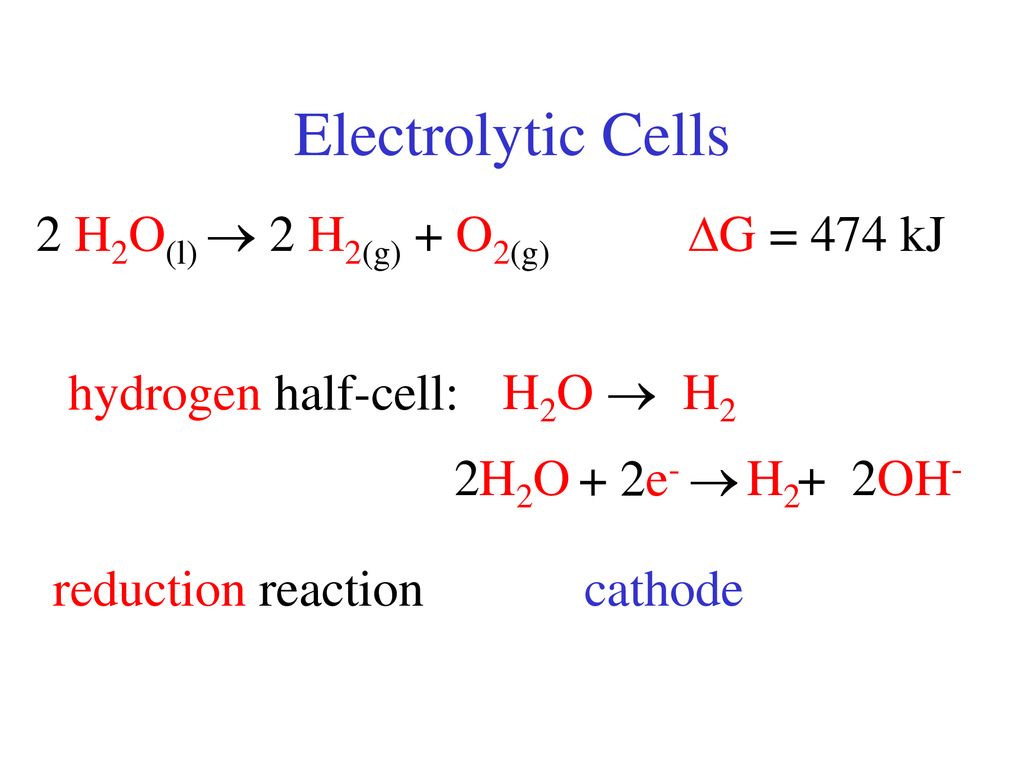
Overall reaction of Cathode and Anode: Cathode: 2 H2O(l) + 2e− → H2(g)... | Download Scientific Diagram
![27) At what concentration of [OH-] does the following half reaction has a potential of 0 V when other species are 1 M? NO3 +H2O+2e - → NO2+ + 2OH"; Eºcell = - 27) At what concentration of [OH-] does the following half reaction has a potential of 0 V when other species are 1 M? NO3 +H2O+2e - → NO2+ + 2OH"; Eºcell = -](https://toppr-doubts-media.s3.amazonaws.com/images/2101378/42282fc5-6d6e-400b-9b95-6369b788e5a7.jpg)
27) At what concentration of [OH-] does the following half reaction has a potential of 0 V when other species are 1 M? NO3 +H2O+2e - → NO2+ + 2OH"; Eºcell = -

Faire-part d'anniversaire H2O, modèle modifiable d'invitation à une fête aquatique H20, invitations de 2e anniversaire Splish Splash, été, enfants, pool party H2B - Etsy France