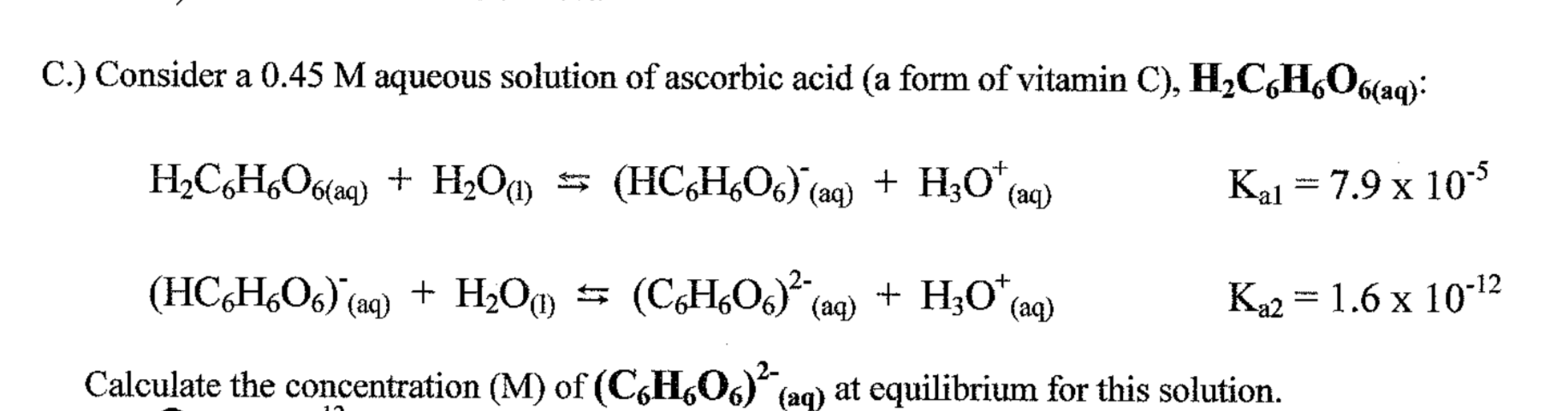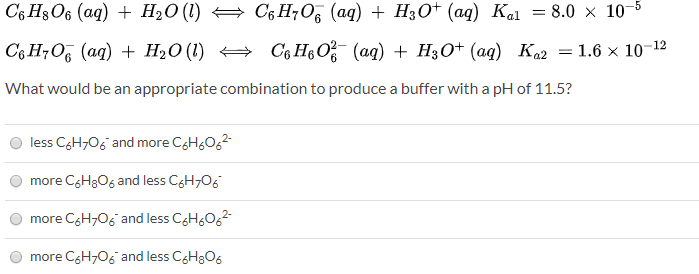SOLVED: The chemical equation for ascorbic acid is: C6H8O6(aq) + H2O(l) â†' C6H7O6-(aq) + H3O+(aq) Change in G = 23.37 kJ What can be determined? The reaction is at equilibrium. The reaction

SOLVED: Texts: A vitamin C (C6H8O6) tablet was dissolved in deionized water and made up to a total volume of 250 mL in a volumetric flask. A 25.00 mL aliquot of this

SOLVED: The chemical equation for ascorbic acid is: C6H8O6(aq) + H2O(l) â†' C6H7O6-(aq) + H3O+(aq) Change in G = 23.37 kJ What can be determined? The reaction is at equilibrium. The reaction

How the Multiple Antioxidant Properties of Ascorbic Acid Affect Lipid Oxidation in Oil-in-Water Emulsions | Journal of Agricultural and Food Chemistry
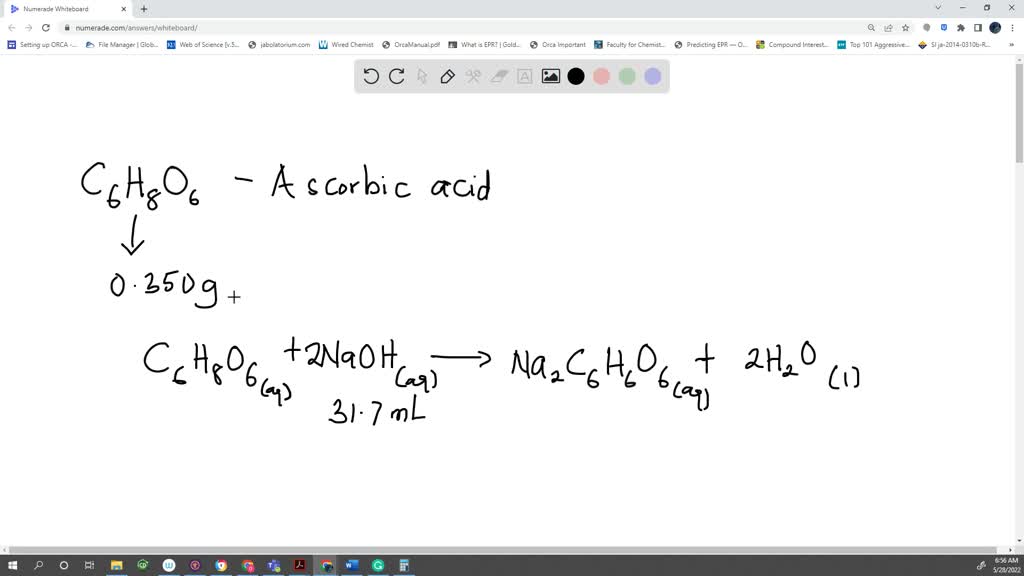
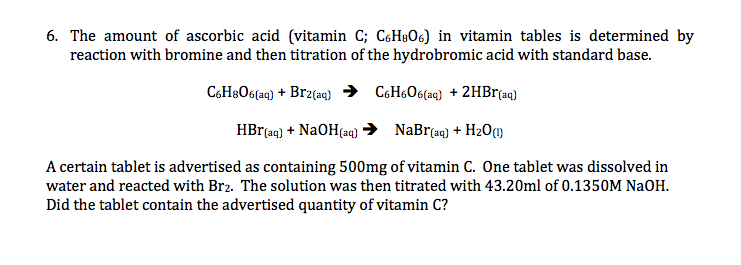
SOLVED: A sample of ascorbic acid, with the formula C6H8O6, is to be studied by titration. First, 0.350 grams of the acid are added to a flask. Sodium hydroxide solution is then
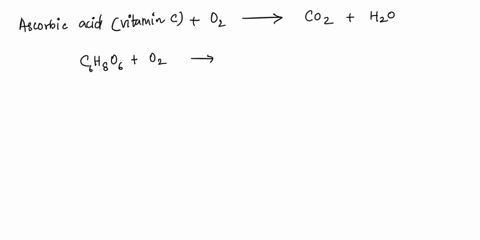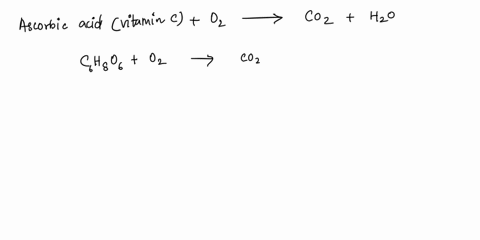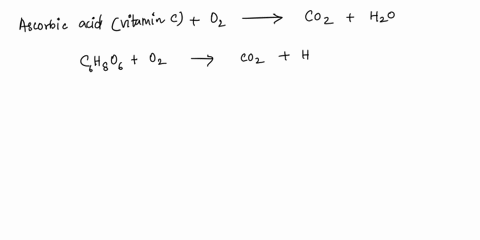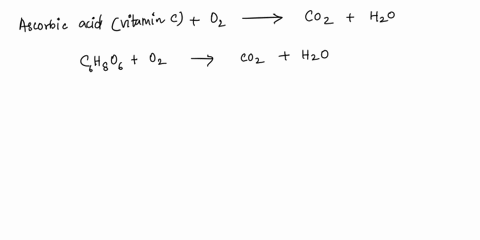
SOLVED: What is the balanced equation for the combustion of ascorbic acid ( C6H8O6, vitamin C) in oxygen to form CO2 and H2O? C6H8O6 + O2 -> CO2 + H2O
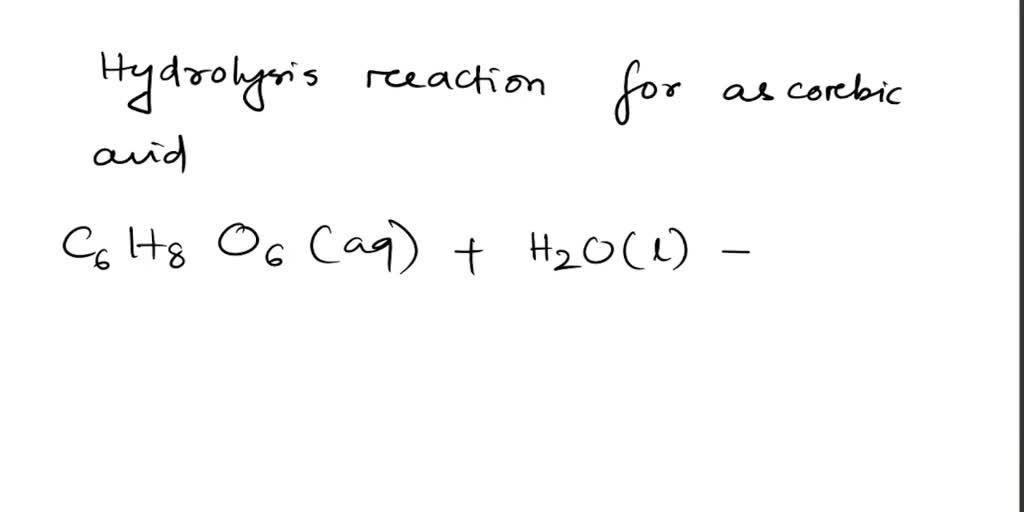
SOLVED: Absorbic Acid (C6H8O6) is a weak acid. a.) Write the hydrolysis reaction for ascorbic acid. b.) Write the acid-dissociation constant (Ka) expression for the reaction in part (a). c.) A 0.100
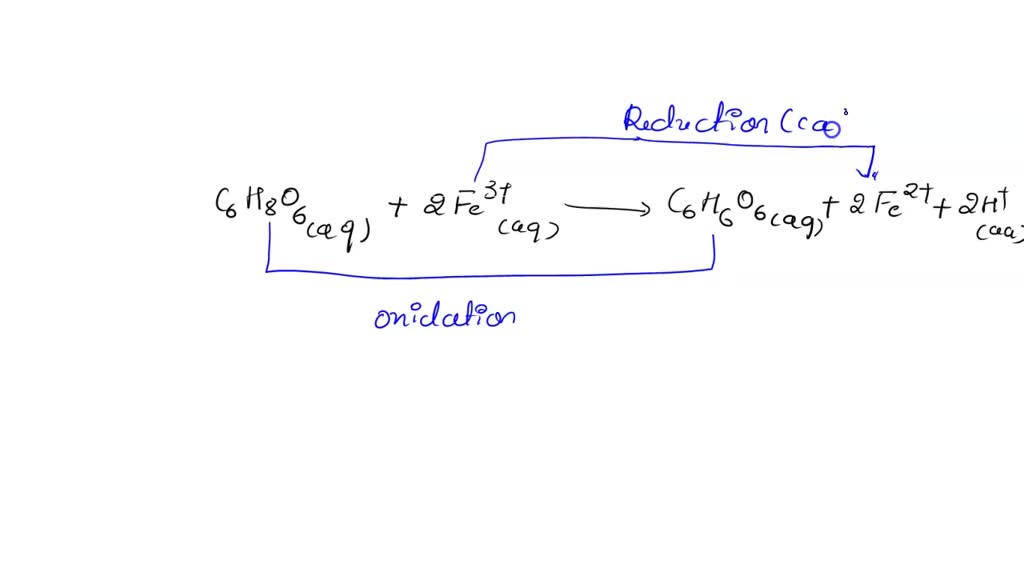
SOLVED: Ascorbic acid (C6H8O6), also commonly known as vitamin C, can be used to reduce a wide variety of transition metal ions. Given that Ecell = 0.71 V for the reaction C6H8O6(aq) +
![SOLVED: Ascorbic acid (C6H8O6) is a diprotic acid. Its Ka values are listed below. What is the [C6H8O6-2] in a 0.15 M solution of ascorbic acid? C6H8O6 (aq) + H2O (l) ⇌ SOLVED: Ascorbic acid (C6H8O6) is a diprotic acid. Its Ka values are listed below. What is the [C6H8O6-2] in a 0.15 M solution of ascorbic acid? C6H8O6 (aq) + H2O (l) ⇌](https://cdn.numerade.com/ask_images/55e213a8c21947539aafd32122cc8bef.jpg)
SOLVED: Ascorbic acid (C6H8O6) is a diprotic acid. Its Ka values are listed below. What is the [C6H8O6-2] in a 0.15 M solution of ascorbic acid? C6H8O6 (aq) + H2O (l) ⇌

SOLVED: Again, consider an aqueous solution of ascorbic acid (Vitamin C, C6H8O6): (1) C6H8O6(aq) + H2O(l) = C6H6O6(aq) + H3O+(aq) Ka1 = 1.0x10^(-1) (2) C6H8O6-(aq) + H2O(l) = C6H6O6^2-(aq) + H3O+(aq) Ka2 =

NCERT Solution Intext 2.11: Calculate the mass of ascorbic acid (Vitamin C, C6H8O6) to be dissolve - YouTube