The experimental enthalpies of solution of Y, BaCO3 and CoCl2·4.24H2O... | Download Scientific Diagram

PPT - What is the difference between a chemical reaction and physical change? PowerPoint Presentation - ID:5813241

Synthetic pathway of 1. a) NH2OH·HCl, BaCO3, Pd/C, N2H4·H2O, reflux in... | Download Scientific Diagram
Calculate the mass of BaCO3 produced when excess CO2 is bubbled through a solution containing 0.205 moles of Ba(OH)2. - Sarthaks eConnect | Largest Online Education Community

Date twe of bor WYSICAL SUENCE Works Balance the following equations: 1. AL + N2 - AIN 2. Fe + 02 - Fe3O4 Caco - CaO + CO2 NH.NO, N2O + H2O

Solve 53 sum 200 m' solution molarity of tho volumo ot CO, at S T P on heat'0 9 (2) - Chemistry - - 12898155 | Meritnation.com

24.Write equilibrium constant expression the following reac (i) BaCO3 (8) ---------- BaO(s) + CO2 (g).
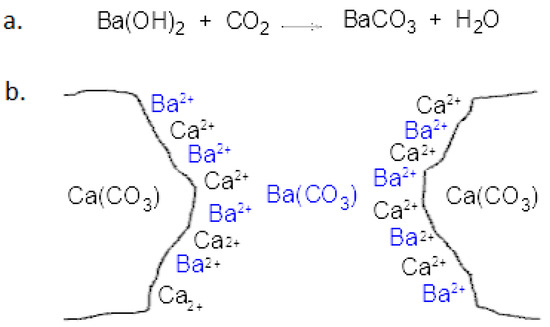
Heritage | Free Full-Text | Characterization of Barium Hydroxide Used as Consolidating Agent for Monumental Surfaces in Venice
Trajectory and timescale of oxygen and clumped isotope equilibration in the dissolved carbonate system under normal and enzymati

Vidéo de question : Identifier la substance qui contient à la fois des liaisons ioniques et des liaisons covalentes | Nagwa

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

a. SO2 + ? → H2SO3 b. ? +H2O → KOH c. ? + ?→ CaCO3 d. CO2 + ? → BaCO3 + H2O e. ? + H2S04 → MgSO4 + H2O f. Fe + HCl– → ? + ? g. Al203 + H2SO4 → ? + H2O h. N
Solved: The chemical equation here describes a reaction between barium carbonate and nitric acid. [algebra]


![ANSWERED] BaCO3 + 2HNO3 → Ba(NO3)2 + CO₂ + H₂O What ... - Organic Chemistry - Kunduz ANSWERED] BaCO3 + 2HNO3 → Ba(NO3)2 + CO₂ + H₂O What ... - Organic Chemistry - Kunduz](https://media.kunduz.com/media/answer/raw/20220422054128757632-4413054.jpg?type=wm)