SOLVED: Consider the reaction: O3 (g) + H2O (g) <—-> H2 (g) + 2 O2 (g) The reverse rate of this reaction is best described as being equal to: Select one: a.

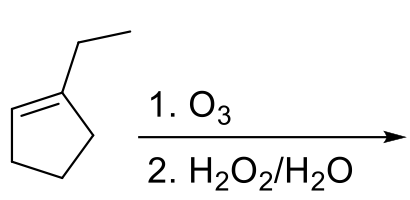
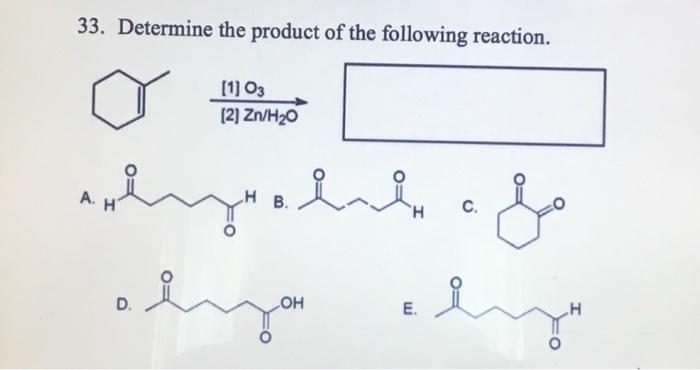
Draw the product formed when the following alkene is treated with O3 followed by Zn, H2O. | Homework.Study.com
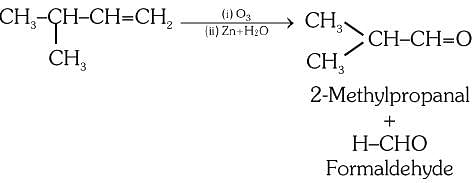
Compound X on reaction with O3 followed by Zn/ H2O gives formaldehyde and 2-methyl propanal as products. The compound X is :a)2-Methylbut-l-eneb)2-Methylbut-2-enec)Pent-2-ened)3-Methylbut-l-eneCorrect answer is option 'D'. Can you explain this answer? -

An alkene "A" on reaction with O3 and Zn–H2O gives propanone and ethanal in equimolar ratio. Addition of HCl - Brainly.in

SOLVED: What are the expected major products from the reaction sequence shown below? 1. O3 2. ZnI2 + H2O CO2 O=C=O OH HO OH O

a) Complete the reactions and identify A, B and C. OH H3PO4 Heat + A+H20 CH3 CH=CH + HBr → B 1 (i) O3 (ii) Zn/H2O C + HCHO (b) Complete the
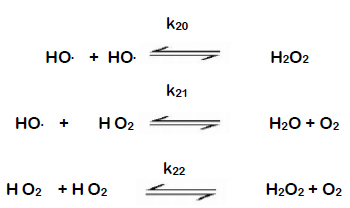
1) H2O2 + O3 → H2O +2O2 2)H2O2 +Ag2O →2Ag +H2O +O2 Determine whether H2O2 is oxidised or reduced in the above reaction? Explain.

An alkene "A" on reaction with O3 and Zn H2O gives propanone and ethanal in equimolar ratio.Addition of HCl to alkene A gives B as the major product. The structure of product
Find out C in the following sequence of reaction cyclohexene +O3 + H20 = A, A +Ca(OH)2 = B, B +C6H5CO3H =
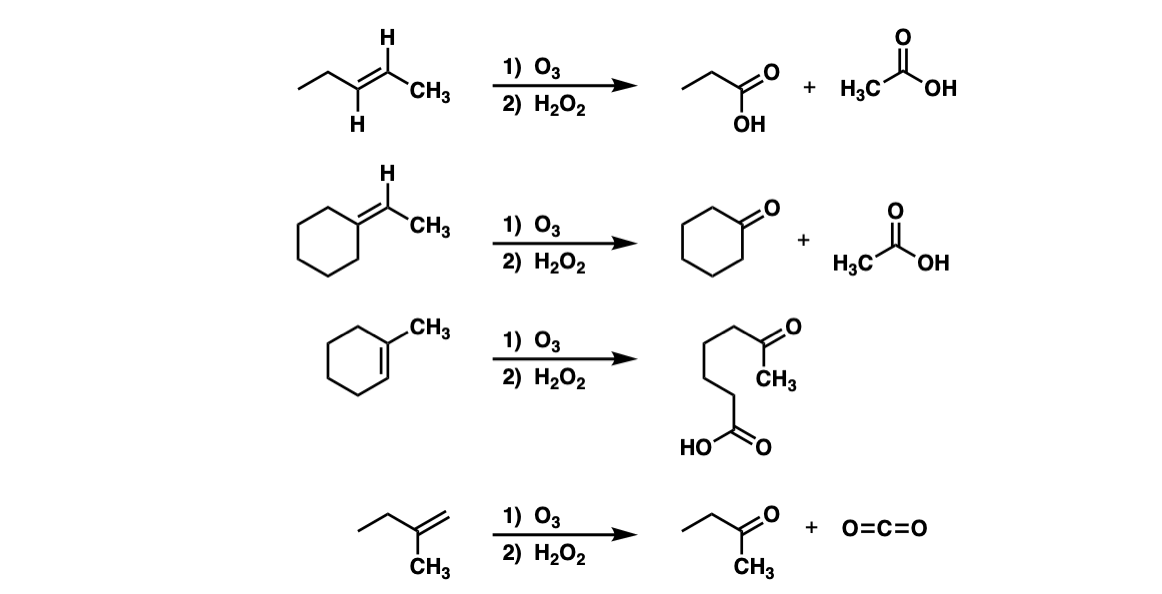
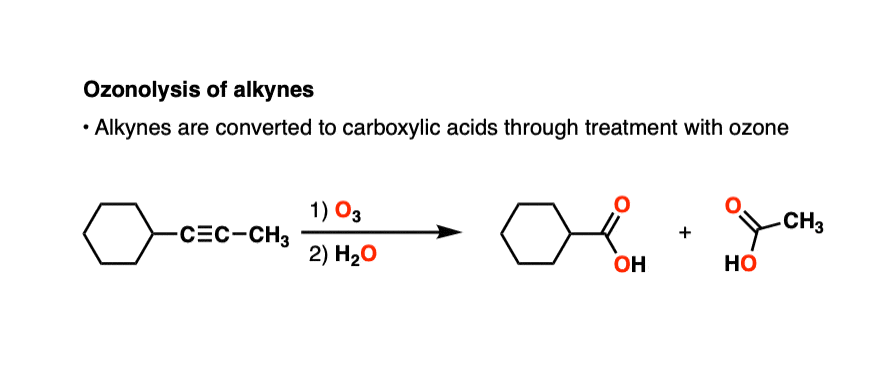
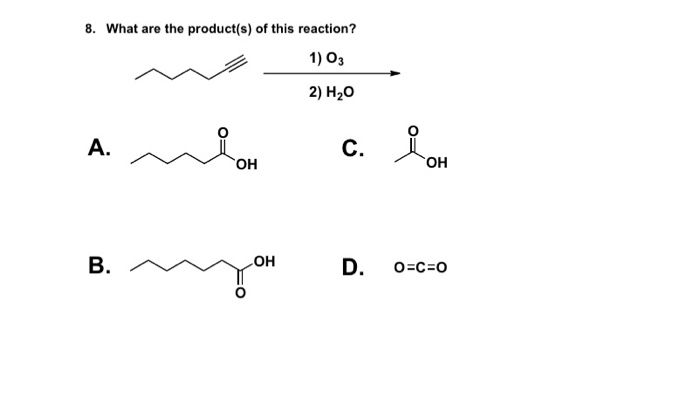
Oxidative cleavage of alkenes to give ketones/carboxylic acids using ozone ( O3) - ("oxidative workup") – Master Organic Chemistry