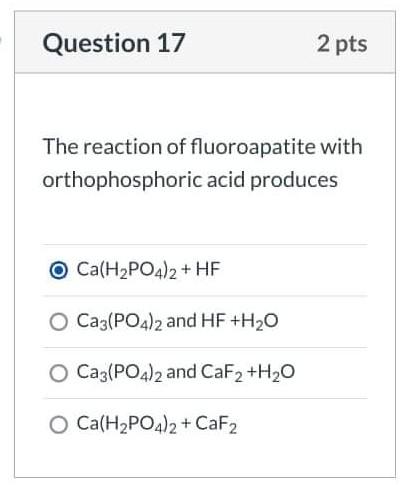
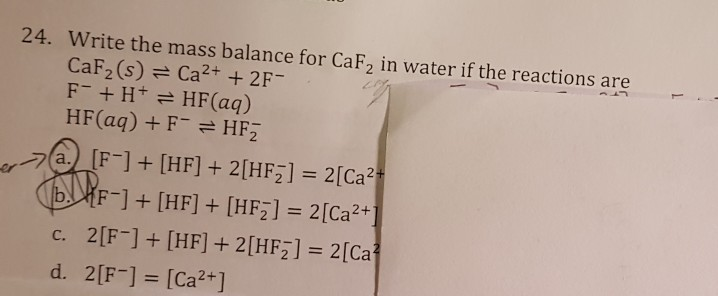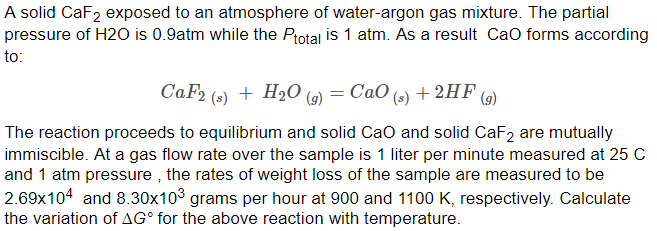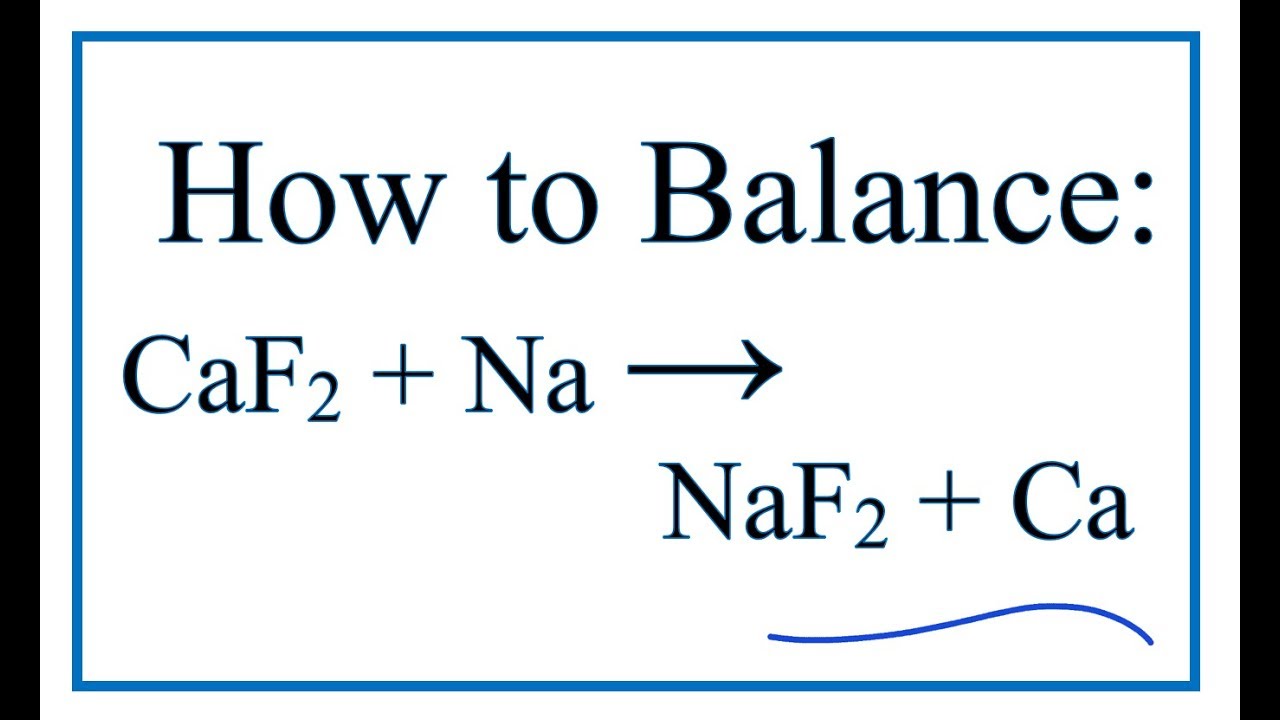SOLVED: Consider the unbalanced chemical equation below: CaSiO3(s) + HF(g) â†' CaF2(aq) + SiF4(g) + H2O(l) Suppose a 34.4 g sample of CaSiO3 is reacted with 31.6 L of HF at 27.0

Charge Reversal Behavior at the CaF2/H2O/SDS Interface as Studied by Vibrational Sum Frequency Spectroscopy | The Journal of Physical Chemistry B

SOLVED: Activity 2: Complex Ion Formation and its Effect on Solubility Consider the two equilibria: CaF2(s) ⇌ Ca2+(aq) + 2F-(aq) Ks = 40 x 10^-11 F-(aq) + H2O(l) ⇌ HF(aq) + OH-(aq)

SOLVED: Which of the following will increase the solubility of calcium fluoride in water? CaF2 (s) = Ca2+ (aq) + 2 F- (aq) Adding calcium nitrate Adding a strong base Adding sodium

Optical Coating Material Calcium Fluoride CaF2 Granule Customized Size China - China Coating Material, Calcium Fluoride | Made-in-China.com